Can I Use a Lithium Battery Instead of Nicd
Batteries are the nearly common ability source for basic handheld devices to large scale industrial applications. A bombardment can be defined as; it is a combination of one or more than electrochemical cells that are capable of converting stored chemic energy into electric energy.

Working of Battery:
A battery is a device, which consists of a various voltaic cells. Each voltaic cell consists of ii one-half cells continued in serial by a conductive electrolyte holding anions and cat ions. One half-prison cell includes electrolyte and the electrode to which anions move, i.e. the anode or negative electrode; the other one-half-jail cell includes electrolyte and the electrode to which cat ions motility, i.e. the cathode or positive electrode.
In the redox reaction that powers the battery, reduction occurs to cations at the cathode, while oxidation occurs to anions at the anode. The electrodes practice not touch one another but are electrically connected past the electrolyte. Mostly the half cells take different electrolytes. All things considered every half-prison cell is enclosed in a container and a separator that is porous to ions but not the bulk of the electrolytes forestall mixing.
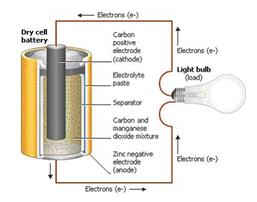
Each half prison cell has an electromotive force (Emf), determined by its capacity to drive electrical electric current from the interior to the exterior of the cell. The net emf of the jail cell is the difference between the emf of its one-half-cells. In this way, if the electrodes take emf and in other words, the net emf is the difference between the reduction potentials of the half-reactions.
How to maintain the Bombardment?
To maintain the battery in good condition, battery equalization is necessary. Due to aging, all the cells do non accuse similarly and some cells accept charge extremely fast while others charge gradually. Equalization can be done by marginally over charging the battery to permit the weaker cells too to accuse completely. The concluding voltage of a completely charged bombardment is12V, automobile battery shows 13.8V in its terminals while a 12 volt tubular battery will testify 14.8V. Automobile battery should be firmly fixed in the vehicle to avert shake. Inverter battery should be placed on a wooden plank if possible.
two Types of Batteries
one) Primary Batteries:
As the proper noun indicates these batteries are meant for single usage. Once these batteries are used they cannot be recharged as the devices are non easily reversible and agile materials may not return to their original forms. Battery manufacturers recommend against recharge of primary cells.
Some of the examples for the disposable batteries are the normal AA, AAA batteries which we use in wall clocks, idiot box remote etc. Other name for these batteries is disposable batteries.


two) Secondary Batteries:
Secondary batteries are as well chosen equally rechargeable batteries. These batteries can be used and recharges simultaneously. They are usually assembled with active materials with active in the discharged land. Rechargeable batteries are recharged by applying electric current, which reverses the chemical reactions that occur during discharge. Chargers are devices which supply the required current.
Some examples for these rechargeable batteries are the batteries used in mobile phones, MP3 players etc. Devices such equally hearing aids and wristwatches use miniature cells and in places such as phone exchanges or computer data heart's, larger batteries are used.

Types of Secondary (rechargeable) Batteries:
SMF, Lead Acid, Li and Nicd
SMF Bombardment:
SMF is asealed maintenance costless battery, designed to offer reliable, consistent and low maintenance power for UPS applications. These batteries can be subject to deep bike applications and minimum maintenance in rural and power deficit areas. These batteries are available from 12V.
In today's informative world, i can't overlook the requirement for battery systems are designed to recover crucial qualified data and information and run basic instrumentations for desired durations. Batteries are required to evangelize instant power. Unreliable and inferior batteries tin can result in the loss of data and equipment shutdowns that tin can cost companies considerable financial losses. Subsequently, the UPS segments calls for the utilization of a reliable and proven bombardment organization.

Lithium (Li) Bombardment:
Nosotros all utilise it in portable devices such as cell phone, a laptop computer or a power tool. The lithium battery has been ane of the greatest achievements in portable power in the terminal decade; with employ of lithium batteries we have been able to shift from black and white mobile to color mobiles with additional features like GPS, email alerts etc. These are the high free energy density potential devices for higher capacities. And relatively low self-discharge batteries. Also Special cells can provide very loftier current to applications such every bit power tools.

Nickel Cadmium (Nicd) Battery:
The Nickel Cadmium batteries have the advantage of beingness recharged many times and possess a relatively constant potential during discharge and take more electrical and concrete withstanding capacity. This battery uses nickel oxide for cathode, a cadmium compound for anode and potassium hydroxide solution as its electrolyte.
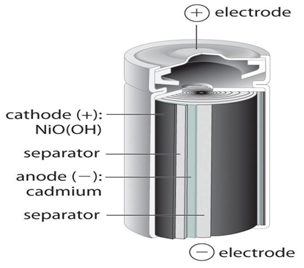
When the battery is charged, the chemical composition of the cathode is transformed and the nickel hydroxide changes to NIOOH. In the anode, formation of Cadmium ions have identify from Cadmium Hydroxide. When battery is discharged, the cadmium reacts with NiOOH to form back nickel hydroxide and Cadmium Hydroxide.
Cd + 2H2O + 2NiOOH —> 2Ni(OH)two + Cd(OH)2
Pb Acid Battery:
Pb Acid batteries are widely used in automobiles, inverters, backup power systems etc. Unlike tubular and maintenance gratis batteries, Atomic number 82 Acrid batteries require proper care and maintenance to prolong its life. The Atomic number 82 Acrid battery consists of a series of plates kept immersed in sulphuric acid solution. The plates have grids on which the agile cloth is attached. The plates are divided into positive and negative plates. The positive plates concur pure atomic number 82 as the active cloth while pb oxide is attached on the negative plates.
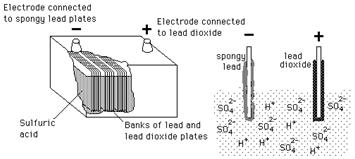
A completely charged bombardment tin can discharge its electric current when connected to a load. During the process of discharge, the sulphuric acid combines with the agile materials on the positive and negative plates resulting in the formation of Lead sulphate. H2o is the single most important stride in maintaining a Lead Acrid bombardment. The frequency of water depends on usage, charge method and operating temperature. During process, the hydrogen atoms from the sulphuric acid react with oxygen to form h2o.
This results in the release of electrons from the positive plates which volition be accustomed by the negative plates. This leads to the formation of an electric potential beyond the battery. The electrolyte in the Pb Acid battery is a mixture of Sulphuric acid and water which has a specific gravity. Specific gravity is the weight of the acid-water mixture compared to equal volume of water. The specific gravity of pure ions complimentary water is i.
The lead-acid batteries provide the best value for power and energy per kilowatt-hour; have the longest life cycle and a large environmental advantage in that they are recycled at an extraordinarily high rate. No other chemistry can touch the infrastructure that exists for collecting, transporting and recycling lead-acid batteries.
Forth with this article, Lithium ion battery is discussed with its advantages and disadvantages.
Working of Lithium – Ion Battery

Lithium –Ion batteries are now popular in majority of electronic portable devices like Mobile phone, Laptop, Digital Camera, etc due to their long lasting power efficiency. These are the near popular rechargeable batteries with advantages like all-time energy density, negligible charge loss and no retentiveness effect. Li-Ion battery uses Lithium ions as the accuse carriers which move from the negative electrode to the positive electrode during discharge and back when charging. During charging, the external current from the charger applies an over voltage than that in the bombardment. This forces the electric current to laissez passer in the reverse management from the positive to the negative electrode where the lithium ions get embedded in the porous electrode fabric through a procedure called Intercalation. The Li- Ions pass through the non aqueous electrolyte and a separator diaphragm. The electrode material is intercalated lithium compound.
The negative electrode of the Li-Ion battery is made upwardly of carbon and the positive electrode is a metal oxide. The almost commonly used material in the negative electrode is Graphite while that in the positive electrode may be Lithium cobalt oxide, Lithium ion phosphate or Lithium manganese oxide. Lithium table salt in an organic solvent is used as the electrolyte. The electrolyte is typically a mixture of organic carbonates like Ethylene carbonate or Diethyl carbonate containing lithium ions. The electrolyte uses anion salts like Lithium hexa fluoro phosphate, Lithium hexa fluoro arsenate monohydrate, Lithium per chlorate, Lithium hexa fluoro borate etc. Depending upon the table salt used, the voltage, capacity and life of the battery varies. Pure lithium reacts with h2o vigorously to form lithium hydroxide and hydrogen ions. So the electrolyte used is non aqueous organic solvent. The electrochemical role of the electrodes charge betwixt anode and cathode depends on the direction of electric current flow.
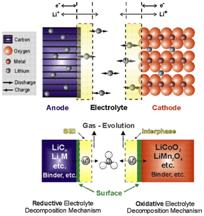
In the Li-Ion battery, both the electrodes tin accept and release lithium ions. During the Intercalation process, the lithium ions move into the electrode. During the reverse process called de intercalation, the lithium ions move back. During discharging, the positive lithium ions will be extracted from the negative electrodes and inserted into the positive electrode. During the charging procedure, the reverse movement of lithium ions takes place.
Advantages of Lithium – Ion Battery:
Lithium Ion batteries outperform NiCd batteries and other secondary batteries. Some of the advantages are
- Light weight compared to other batteries of similar size
- Available in different shape including Flat shape
- High open excursion voltage that increases the power transfer at low electric current
- Lack of retentiveness effect.
- Very depression self discharge rate of v-10% per month. Self discharge is effectually xxx% in NiCd and NiMh batteries.
- Eco-friendly bombardment without whatever free lithium metallic
Just along with the advantages, like other batteries, Li-Ion battery also suffers from some disadvantages.
Disadvantages of Li-Ion Battery:
- The deposits inside the electrolyte over time will inhibit the flow of charge. This increases the internal resistance of the bombardment and the cell's capacity to deliver current gradually decreases.
- High charging and loftier temperature may leads to capacity loss
- When overheated, Li-Ion battery may suffer thermal run abroad and cell rupture.
- Deep discharge may short circuit the Li-Ion battery. Then to prevent this, some makes have internal close down circuitry that shut down the battery when its voltage is higher up the safe level of 3 to 4.2 volts. In this example, when the battery is not using for long time, the internal circuitry volition consume ability and drain the battery below its shut down voltage. So to charge such batteries normal chargers are non useful.
armstrongsaidgety.blogspot.com
Source: https://www.elprocus.com/batteries-types-working/
0 Response to "Can I Use a Lithium Battery Instead of Nicd"
Post a Comment